Devices and Diagnostics
PODCAST EPISODE 315 Click here to watch the video version of this podcast. Retina specialists have limited imaging and surgical tools to visualize the…
Read MoreEven the most groundbreaking device won’t go far if doctors can’t easily adopt it into their practice. PODCAST EPISODE 306
Read MoreJudy Gordon, DVM, is not a job-hopper. When Laguna Beach, Calif.-based ClinReg Consulting Services, a company she founded in 1998, announced her retirement in March,…
Read MoreFirst-in-class drugs are being investigated to treat two etiologies of dry eye disease (DED)—meibomian gland dysfunction (MGD) and Demodex blepharitis—both highly prevalent conditions for which…
Read MoreThe ocular allergy space has seen some renewed interest lately, with Food and Drug Administration approval of Santen Pharmaceutical’s new drug for vernal keratoconjunctivitis (VKC)…
Read MoreIn the summer of 2020, shortly after the US Food and Drug Administration issued a negative Complete Response Letter to AbbVie and Molecular Partners, rejecting…
Read MoreUp until a few weeks ago, 2021 had been an up-and-down year for retinal prosthesis maker Second Sight Medical Products, but it recently got a…
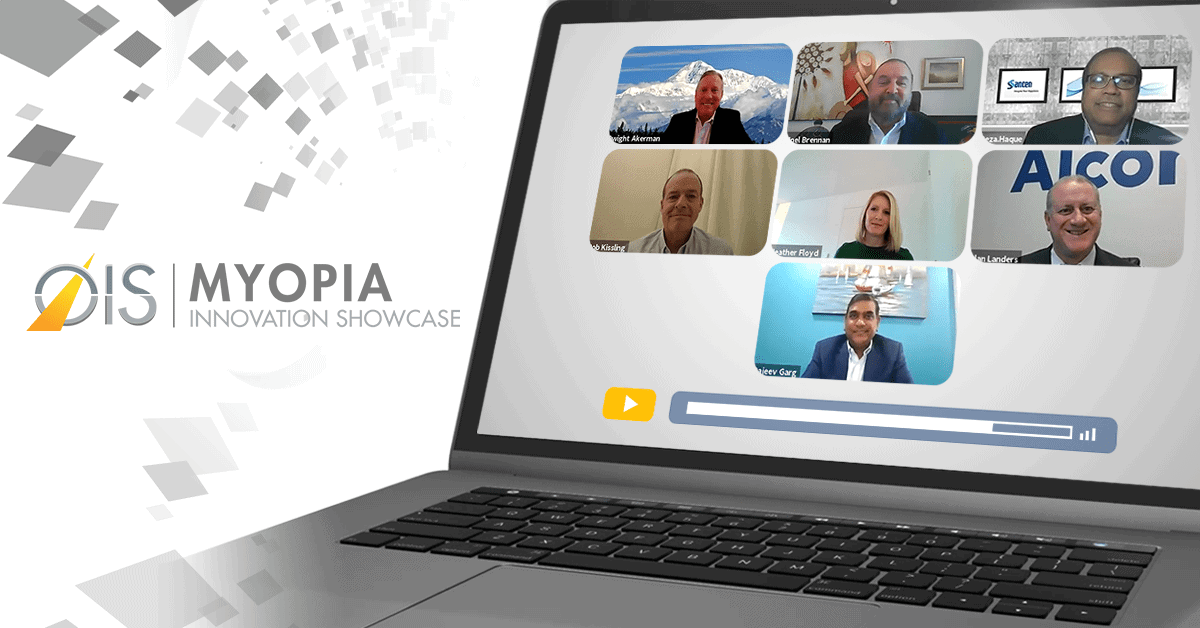
Read More“How big is big?” That was a question that Dwight Akerman, OD, MBA, FAAO, FBCLA, put to a panel of industry insiders at last week’s…
Read MoreThe glaucoma space has been dominated by a host of products designed to lower intraocular pressure (IOP) – minimally invasive glaucoma surgery (MIGS) devices and…
Read MoreSUBSCRIBE TO OIS NEWS
Get Our Weekly Newsletter About The Latest In Ophthalmic Innovation.
We respect your privacy and promise to never send you spam.













![OIS-ANTERIOR-NEW-DATE-750x420[4300] OIS-ANTERIOR-NEW-DATE-750x420[4300]](https://ois.net/wp-content/uploads/2020/03/OIS-ANTERIOR-NEW-DATE-750x4204300.jpg)